Toxins | Free Full-Text | Membrane Permeabilization by Pore-Forming RTX Toxins: What Kind of Lesions Do These Toxins Form? | HTML

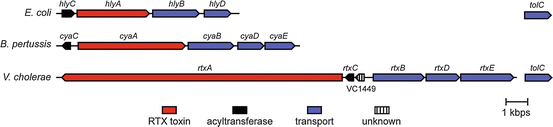
Identification of a Vibrio cholerae RTX toxin gene cluster that is tightly linked to the cholera toxin prophage | PNAS

Bacterial RTX Toxins Allow Acute ATP Release from Human Erythrocytes Directly through the Toxin Pore* - Journal of Biological Chemistry

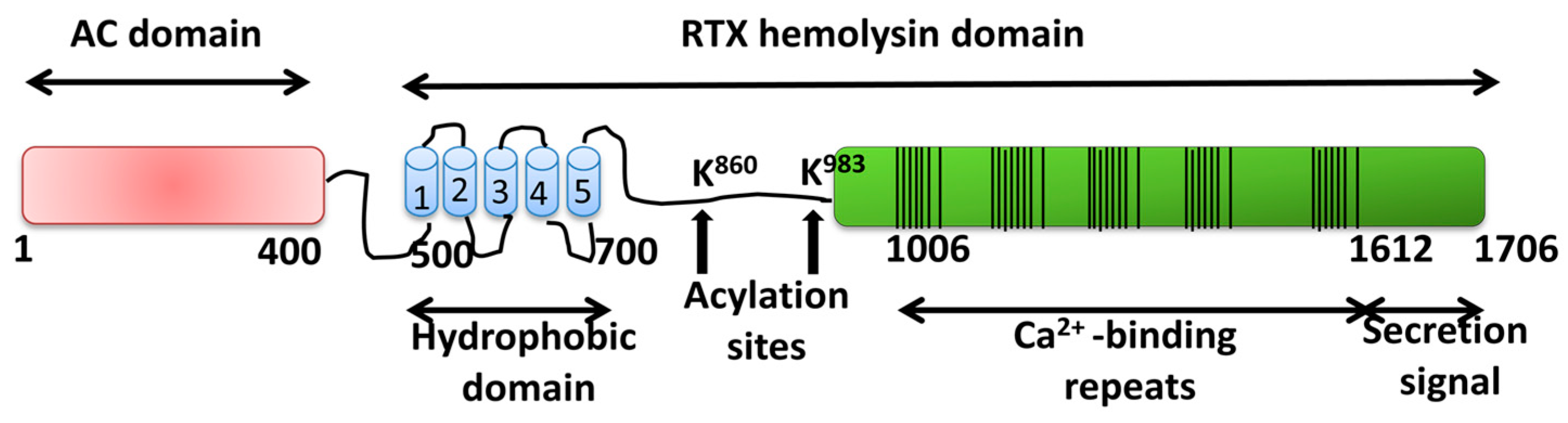
The RTX pore-forming toxin α-hemolysin of uropathogenic Escherichia coli: progress and perspectives | Future Microbiology

His-859 Is an Essential Residue for the Activity and pH Dependence of Escherichia coli RTX Toxin α-Hemolysin* - Journal of Biological Chemistry

2 Schematic organization of HlyA and CyaA RTX toxins. The different... | Download Scientific Diagram

RCSB PDB - 5CXL: CRYSTAL STRUCTURE OF RTX DOMAIN BLOCK V OF ADENYLATE CYCLASE TOXIN FROM BORDETELLA PERTUSSIS

Pore forming activity of the potent RTX-toxin produced by pediatric pathogen Kingella kingae: Characterization and comparison to other RTX-family members - ScienceDirect

Continuous Assembly of β-Roll Structures Is Implicated in the Type I-Dependent Secretion of Large Repeat-in-Toxins (RTX) Proteins - ScienceDirect

In vivo covalent cross‐linking of cellular actin by the Vibrio cholerae RTX toxin | The EMBO Journal

Vibrio cholerae Strains with Mutations in an Atypical Type I Secretion System Accumulate RTX Toxin Intracellularly | Journal of Bacteriology
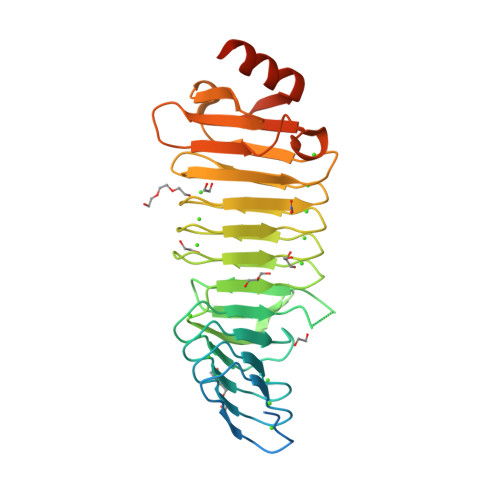
RCSB PDB - 6SUS: Crystal structure of RTX domain blocks IV and V of adenylate cyclase toxin from Bordetella pertussis

RTX proteins: a highly diverse family secreted by a common mechanism - Linhartová - 2010 - FEMS Microbiology Reviews - Wiley Online Library

Coordinated delivery and function of bacterial MARTX toxin effectors - Woida - 2018 - Molecular Microbiology - Wiley Online Library
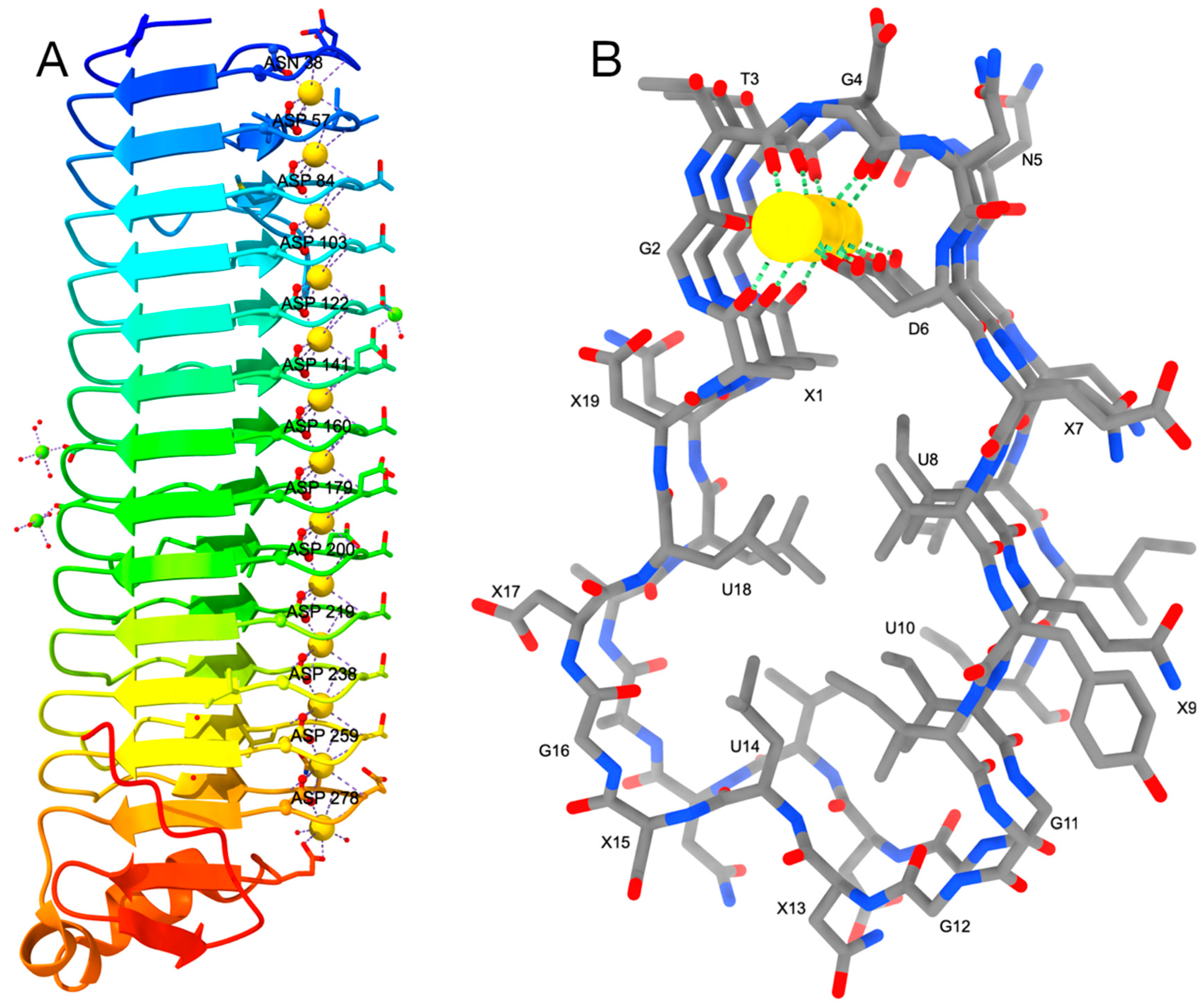
Toxins | Free Full-Text | Structure–Function Relationships of the Repeat Domains of RTX Toxins | HTML

Figure 2 from Structure and function of MARTX toxins and other large repetitive RTX proteins. | Semantic Scholar

Almost half of the RTX domain is dispensable for complement receptor 3 binding and cell-invasive activity of the Bordetella adenylate cyclase toxin - Journal of Biological Chemistry