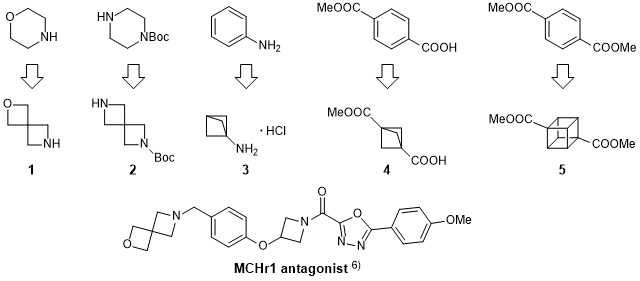
Ring Expansion of 3‐Oxetanone‐Derived Spirocycles: Facile Synthesis of Saturated Nitrogen Heterocycles - Ruider - 2013 - Angewandte Chemie International Edition - Wiley Online Library
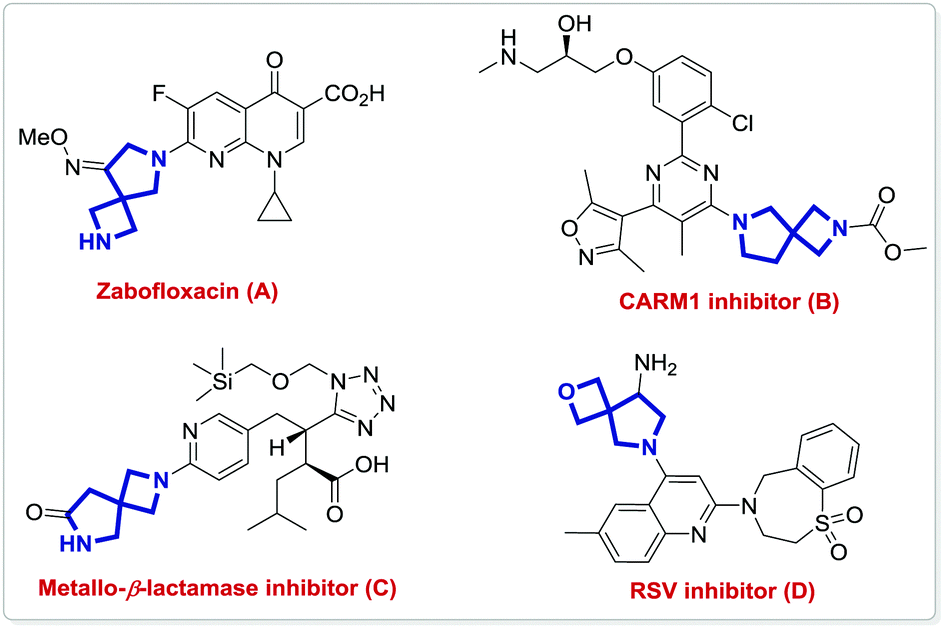
Ligand-controlled switch in diastereoselectivities: catalytic asymmetric construction of spirocyclic pyrrolidine-azetidine/oxe(thie)tane derivatives - Chemical Communications (RSC Publishing)

Catalysts | Free Full-Text | Gold-Catalyzed Synthetic Strategies towards Four-Carbon Ring Systems | HTML

Mild C–C Bond Formation via Lewis Acid Catalyzed Oxetane Ring Opening with Soft Carbon Nucleophiles - Huang - 2021 - Angewandte Chemie - Wiley Online Library

PDF) Spirocyclic Oxetanes: Synthesis and Properties | Isabelle Parrilla and Mark Rogers-evans - Academia.edu
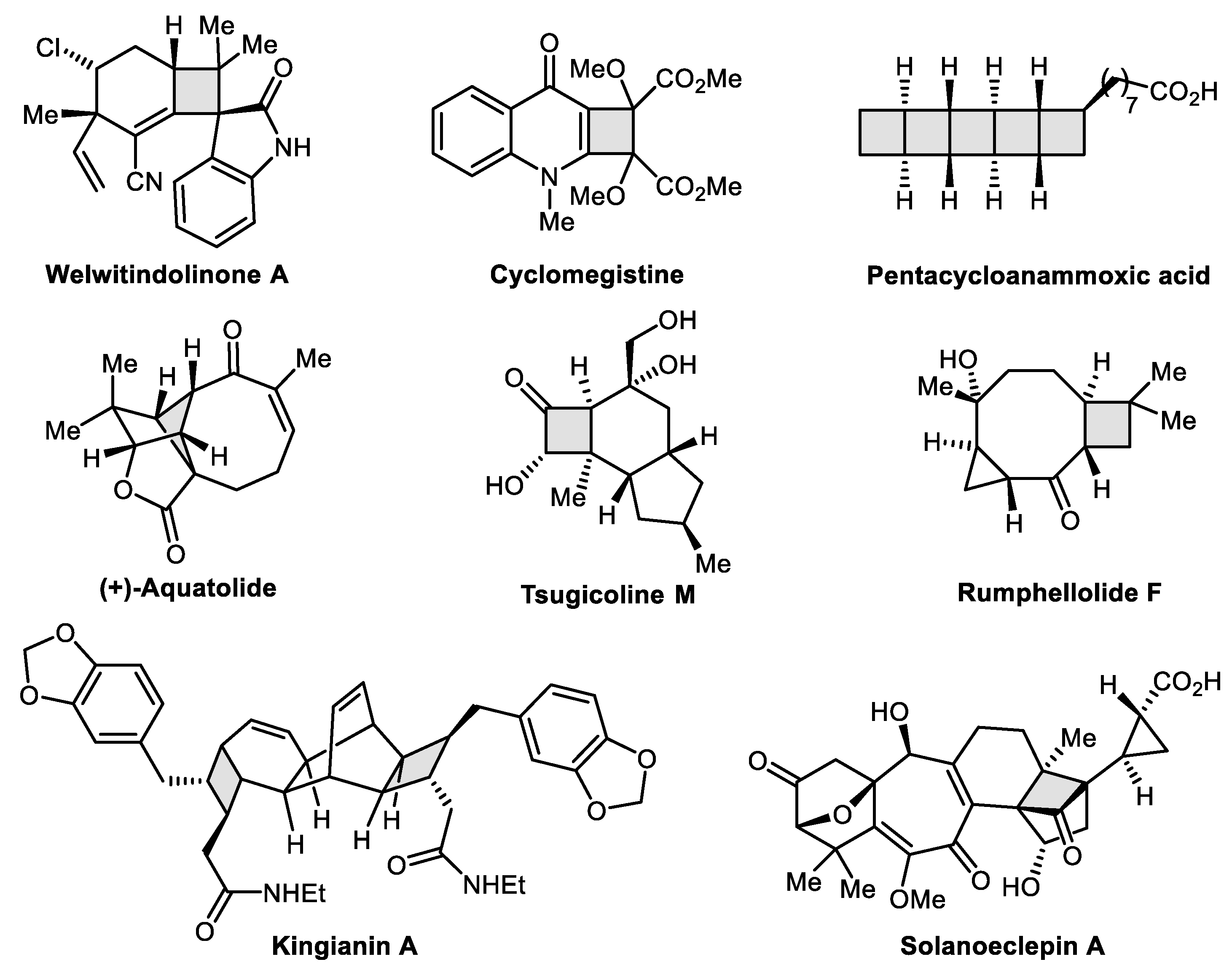
Catalysts | Free Full-Text | Gold-Catalyzed Synthetic Strategies towards Four-Carbon Ring Systems | HTML

Deutschland über Alles: Thomas Grey on Richard Wagner's Musical Genius, Nationalism, and Controversial Legacy - Simply Charly
![Catalytic Asymmetric Synthesis of 2‐Alkyleneoxetanes through [2+2] Annulation of Allenoates with Trifluoromethyl Ketones - Zhao - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library Catalytic Asymmetric Synthesis of 2‐Alkyleneoxetanes through [2+2] Annulation of Allenoates with Trifluoromethyl Ketones - Zhao - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/5f8ddb21-2ca3-4672-8a1e-818958283c04/mcontent.jpg)








![NY Times Article] The Stakes Are Sky-High for a 'Ring' Coming to the Met Opera : r/opera NY Times Article] The Stakes Are Sky-High for a 'Ring' Coming to the Met Opera : r/opera](https://external-preview.redd.it/ltk3sKplXDzhmjXClWeXlSDJX8Rf4FcSZGOIyV5rgkY.jpg?auto=webp&s=c53a72d1833d526d796423d401426b8a46b1482d)