The Formation of the Combination Products Policy Council to Improve Regulatory Efficiency - BioTechLogic, Inc

How To Apply QbD to Drug Device Combination Products – Quality by Design for Biotech, Pharmaceutical and Medical Devices

2Exceed IFF's ERFA gruppe for combination products Risk Management 15. sept /Ingrid Malmberg. - ppt download

Product Development and Manufacturing Challenges for Combination Products | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Notice: Human Drugs: Drug Products Withdrawn From Sale For Reasons Other Than Safety or Effectiveness— Combination Products and Mutually Conforming Labeling Cross Labeling | PDF | Institutional Review Board | Federal Food

1 Cross Labeling Combination Products and User Fees Bradley Merrill Thompson, MBA, JD, RAC Epstein Becker & Green PC Pharma MedDevice 2007 Conference New. - ppt download
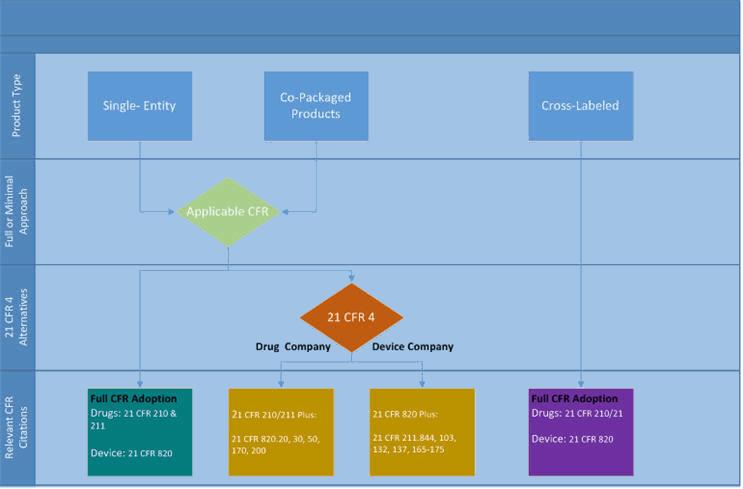
![3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses 3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses](https://learngxp.com/wp-content/uploads/2020/01/ELM-145.png)
3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses
![3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses 3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses](https://learngxp.com/wp-content/uploads/2020/02/ELM-145-02-3-Types-of-Combination-Products-You-Should-Know.png)
3 Types of Combination Products You Should Know [Video] - LearnGxP: Accredited Online Life Science Training Courses

PPT - Combination Products: Cross Labeling and Single Entity Labeling PowerPoint Presentation - ID:726338