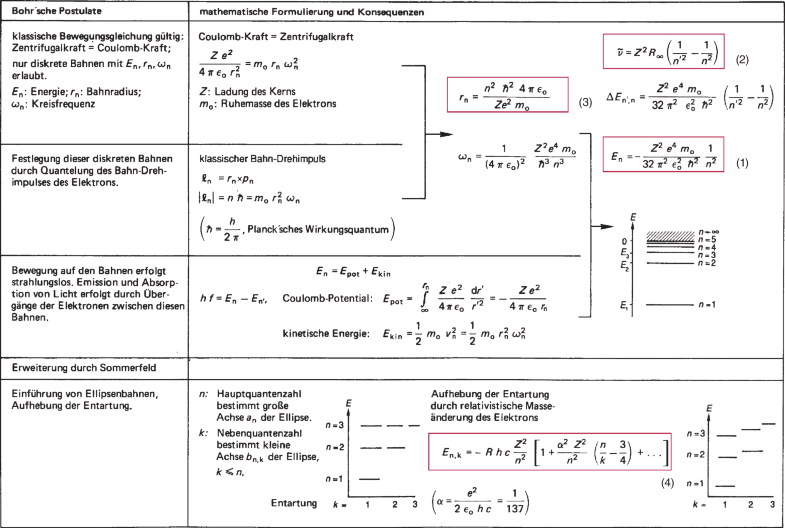
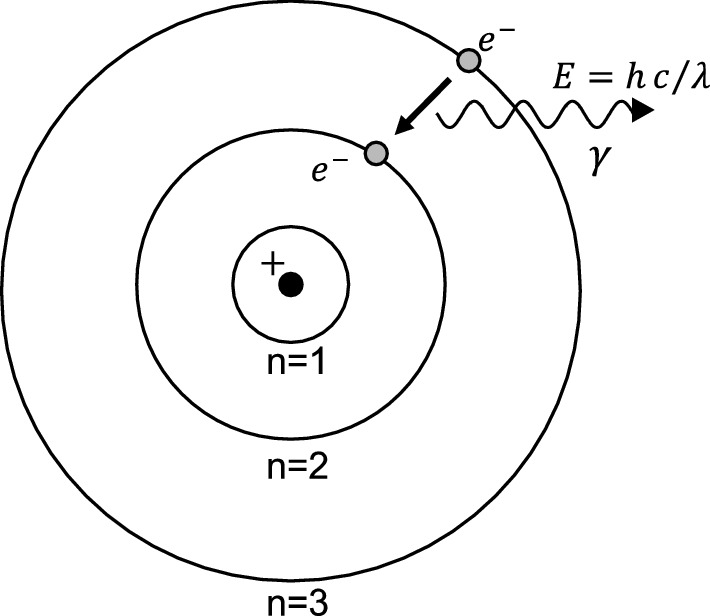
State Bohr's postulate of hydrogen atom which successfully explains the emission lines in the spectrum of

AS) 3. Give the main postulates of Bohr's model of an atom. (AS) Ctate the valencies of magnesium and sodium (AS)

PDF) Collapse or no collapse? What is the best ontology of quantum mechanics in the primitive ontology framework?

Advantages of Bohr’s Theory, Bohr Quantization Postulate, Chemistry Study Material @eMedicalprep.com | eMedicalPrep