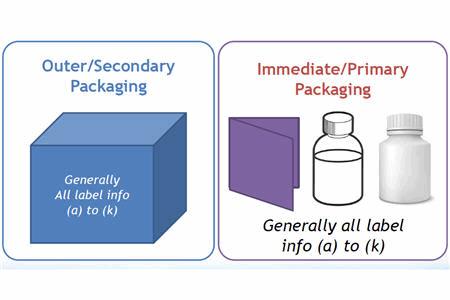
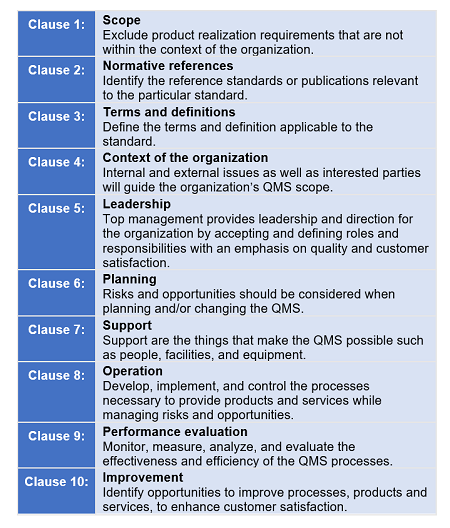
Good Practice Guide: Packaging, Labeling, & Warehousing Facilities | ISPE | International Society for Pharmaceutical Engineering
![Medical Device Labeling - Institute of Packaging ... - IoPP meets the GMP device master record requirements with respect to legibility, adhesion, - [PDF Document] Medical Device Labeling - Institute of Packaging ... - IoPP meets the GMP device master record requirements with respect to legibility, adhesion, - [PDF Document]](https://demo.fdocuments.in/img/378x509/reader019/reader/2020032919/5aa812c57f8b9aac258b5efe/r-2.jpg)
Medical Device Labeling - Institute of Packaging ... - IoPP meets the GMP device master record requirements with respect to legibility, adhesion, - [PDF Document]